|
They were given the name halogen, from the Greek roots hal- (salt) and -gen (to produce), because they all produce sodium salts of similar properties, of which. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). This element, which was first discovered in the sun, was assumed to be an alkali metal, but when it was discovered terrestrially it was found to be an inert gas. halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. Examples are cadmium, lanthanum, lithium, thallium, radium. All metallic elements discovered after this date ( $1811$) have been given names ending in - ium (occasionally - um). The names sodium and potassium were changed to natrium and kalium. He used the Latin names ferrum, stannum and stibium for iron, tin and antimony, and gave zinc the "Latin" name zincum. Key Takeaways: List of the Elements H - Hydrogen He - Helium Li - Lithium Be - Beryllium B - Boron C - Carbon N - Nitrogen O - Oxygen F - Fluorine Ne - Neon Na - Sodium Mg - Magnesium Al - Aluminum, Aluminium Si - Silicon P - Phosphorus S - Sulfur Cl - Chlorine Ar - Argon K - Potassium Ca - Calcium. Some were changed (albeit temporarily), and to some were simply added the suffix.Ī number of names already given to metallic elements, e.g., uranium, chromium, barium, were of this form. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. Some elements had optained such a name prior to $1811$. The Royal Society of Chemistry's interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. The Swedish chemist Berzelius, who recognised the value of logical naming, proposed (in $1811$) that the names of the elements should all be Latin in form. The deliberate naming of chemical elements such that they end in - um or - ium originates in $1811$. 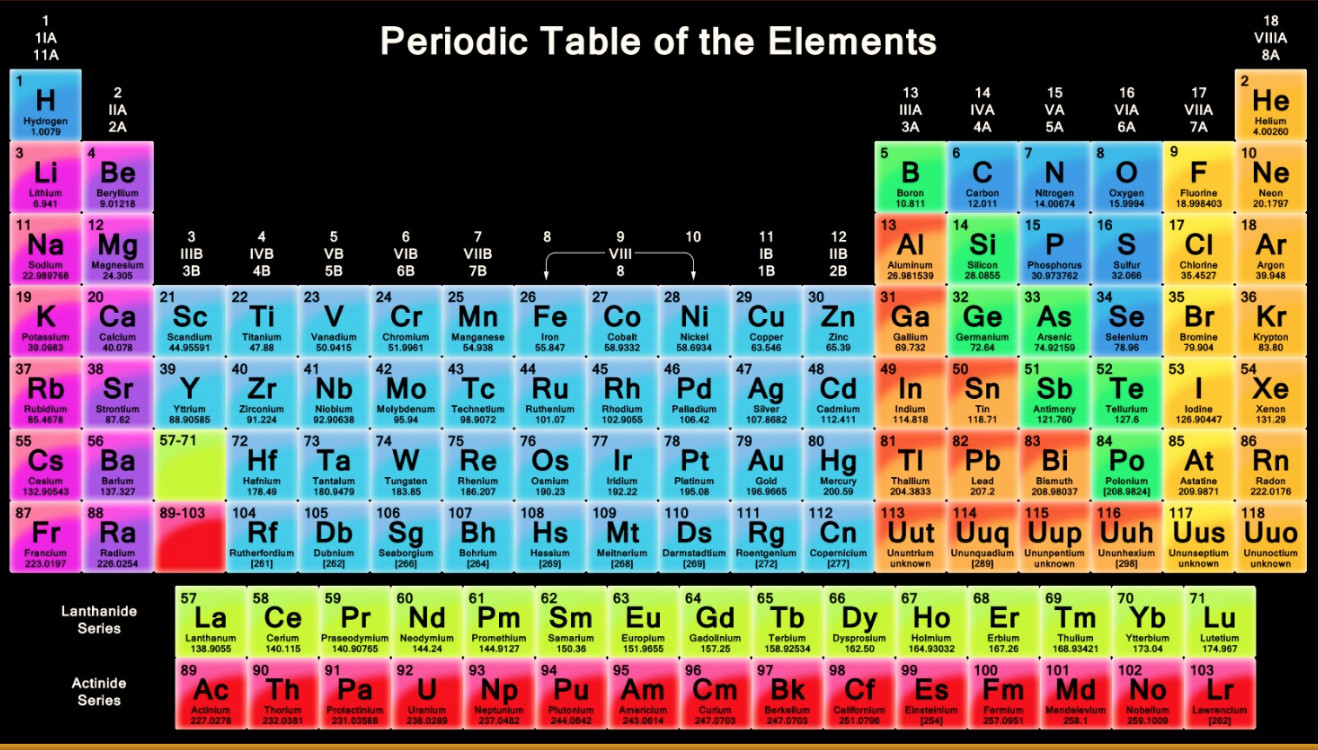
To confirm and add a reference to the other two answers. gold as opposed to aurum) simply by force of usage. List of elements Atomic Number Name Symbol Group Period Number Block State at. So, I think after that, other elements were simply given the suffix to fit the generally useful naming scheme, and then, metal names which were already in common use kept their common language names ( e.g. aurum, argentum, ferrum the names of sodium, potassium, and magnesium, derived from soda, potassa or potash, and magnesia, were given by Davy in 1807, with the derivative form -ium and although some of the later metals have received names in -um, the general form is in -ium, as in cadmium, iridium, lithium, osmium, palladium, rhodium, titanium, uranium in conformity with which aluminum has been altered to aluminium. 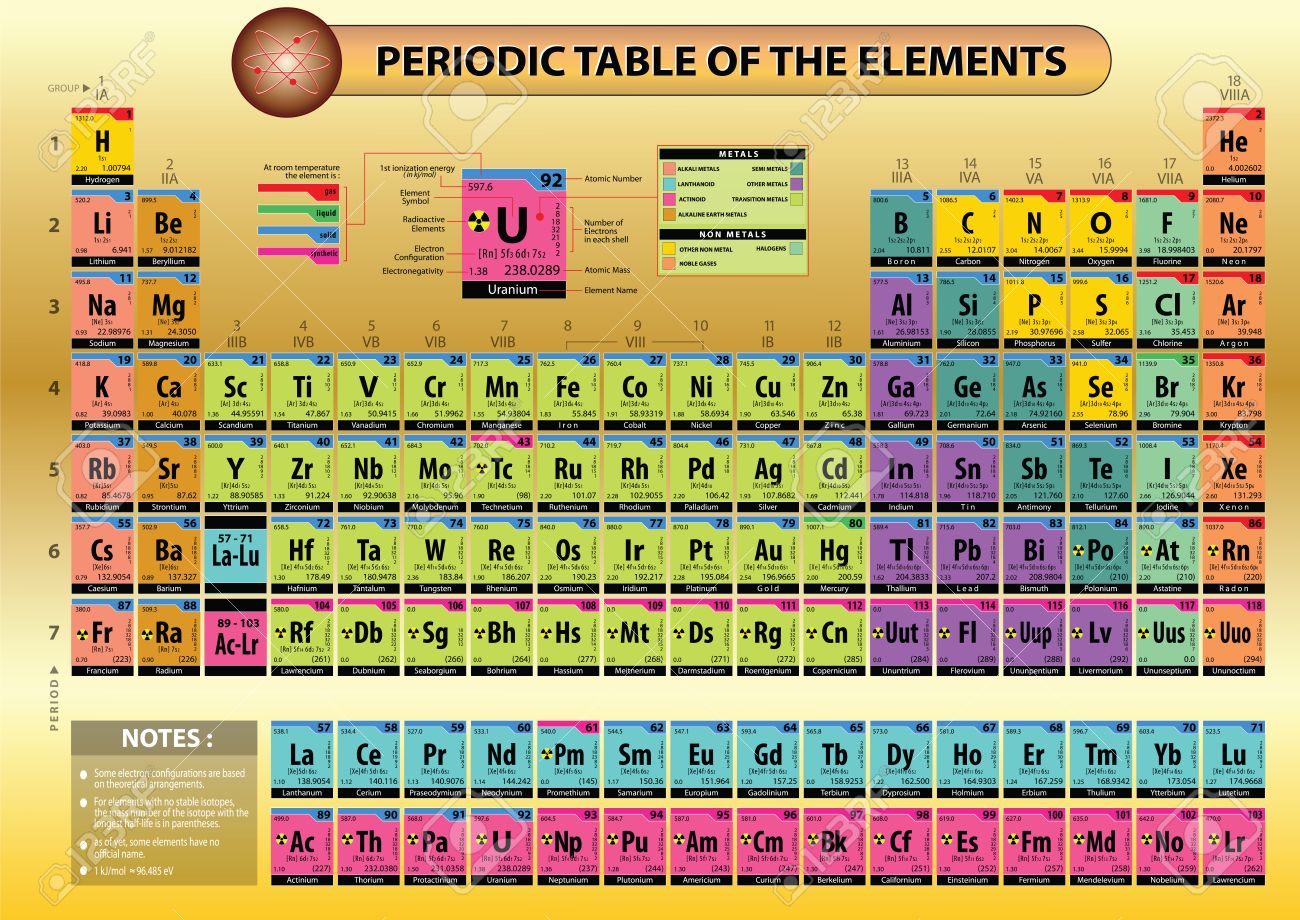
Since Latin didn't have words for the new metallic elements, new words were coined from the existing terms for the substances and given Latinate endings.įrom the OED's entry on -ium: The Latin names of metals were in -um, e.g. 
To expand on answer - in the early 19th century, when the Royal Society was really in the swing of things, the dominant language of scholarship was still Latin.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
